|
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. Method 2 is probably mathematically simpler, but Method 1 makes it clear that you are determining an average mass.For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. No single carbon atom has a mass of 12.01 amu. This is the average atomic mass of carbon. 3 Using the atomic mass of an element and multiplying it by the conversion factor grams per mole (g/mol), you can calculate the molar mass of that element. Molar mass is the mass (in grams) of one mole of a substance. The two methods are mathematically equivalent. On the periodic table the mass of carbon is reported as 12.01 amu. Method 1 Calculating the Molar Mass of an Element 1 Understand molar mass. #"Average mass" = "120 110 u"/"10 000 atoms" = "12.011 u"#Īnother way of determining the average mass is to multiply the atomic mass of each isotope by its percentage and then add the numbers. Its just a very big number because it takes a lot of amus to weigh one gram. The mole is a conversion factor to convert from amus to grams. #"Total mass"color(white)(l)color(white)(mmmmmmmmmmmmm) = "120 110 u"# The atomic masses of 12C and 13C, for example, are 12 Da and 13.003355 Da, respectively, while the atomic weight of carbon is 12.011 Da, which accounts for both. ( 694 votes) Upvote Flag Alf Lyle 10 years ago Absolutely correct. Then you have 9893 atoms of 12C and 107 atoms of 13C. Assume that you have, say, 10 000 atoms of carbon. #"Mass of"color(white)(l) ""^12"C" = 9893 color(red)(cancel(color(black)("atoms"))) × "12 u"/(1 color(red)(cancel(color(black)("atom")))) = "118 716 u"# METHOD 1 To find the average atomic mass, you take a certain number of atoms, find the total mass of each isotope, and then divide the total mass of all the atoms by the total number of atoms. However, the 'unit' Dalton is still used in common practice. Relative atomic and molecular mass values as defined are dimensionless. To find the average atomic mass, you take a certain number of atoms, find the total mass of each isotope, and then divide the total mass of all the atoms by the total number of atoms.Īssume that you have, say, 10 000 atoms of carbon. Atomic and molecular masses are usually reported in daltons which is defined relative to the mass of the isotope 12 C (carbon 12).  
Carbon-12 is composed of 6 protons, 6 neutrons, and 6 electrons. What is the average atomic mass of these isotopes? Carbon-12 is of particular importance in its use as the standard from which atomic masses of all nuclides are measured, thus, its atomic mass is exactly 12 daltons by definition. #""^14"C"# exists only in trace amounts, so it makes no measurable contribution to the average atomic mass.Ĭarbon consists of 98.93% #""^12"C"# (12 u) and 1.07% #""^13"C"# (13.003 u). This second definition is actually the relative atomic mass of an element - a single average value of the element's mass based on the masses of its isotopes.Ĭarbon has 15 known isotopes, of which only two ( #""^12"C"color(white)(l) "and"color(white)(l) ""^13"C"#) are stable. 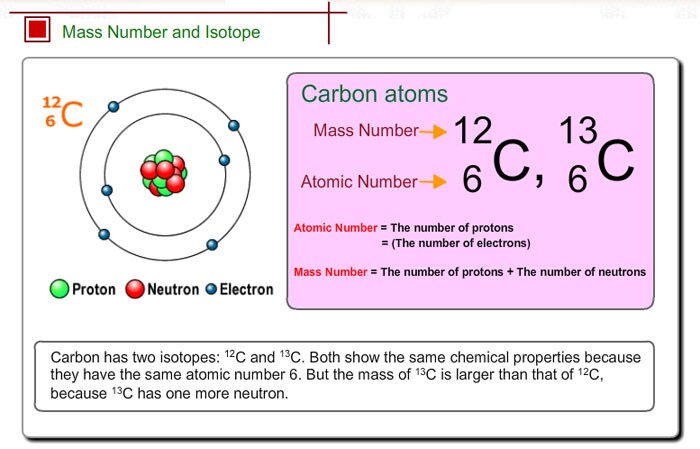
The term atomic mass is also often used (though technically, incorrectly) to refer to the average atomic mass of all of the isotopes of an element. The mass of a single atom of carbon-12 is defined as exactly 12 u. Molecular weight calculation: 12.010712 + 1.0079422 + 15.999411 Percent composition by element Element: Hydrogen Symbol: H Atomic Mass: 1.00794 of Atoms: 22 Mass Percent: 6.478 Element: Carbon Symbol: C Atomic Mass: 12.0107 of Atoms: 12 Mass Percent: 42.106 Element: Oxygen Symbol: O Atomic Mass: 15. The term " atomic mass" refers to the mass of a single atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
